
Uranium: What is it and How does it work?
What is Uranium ?
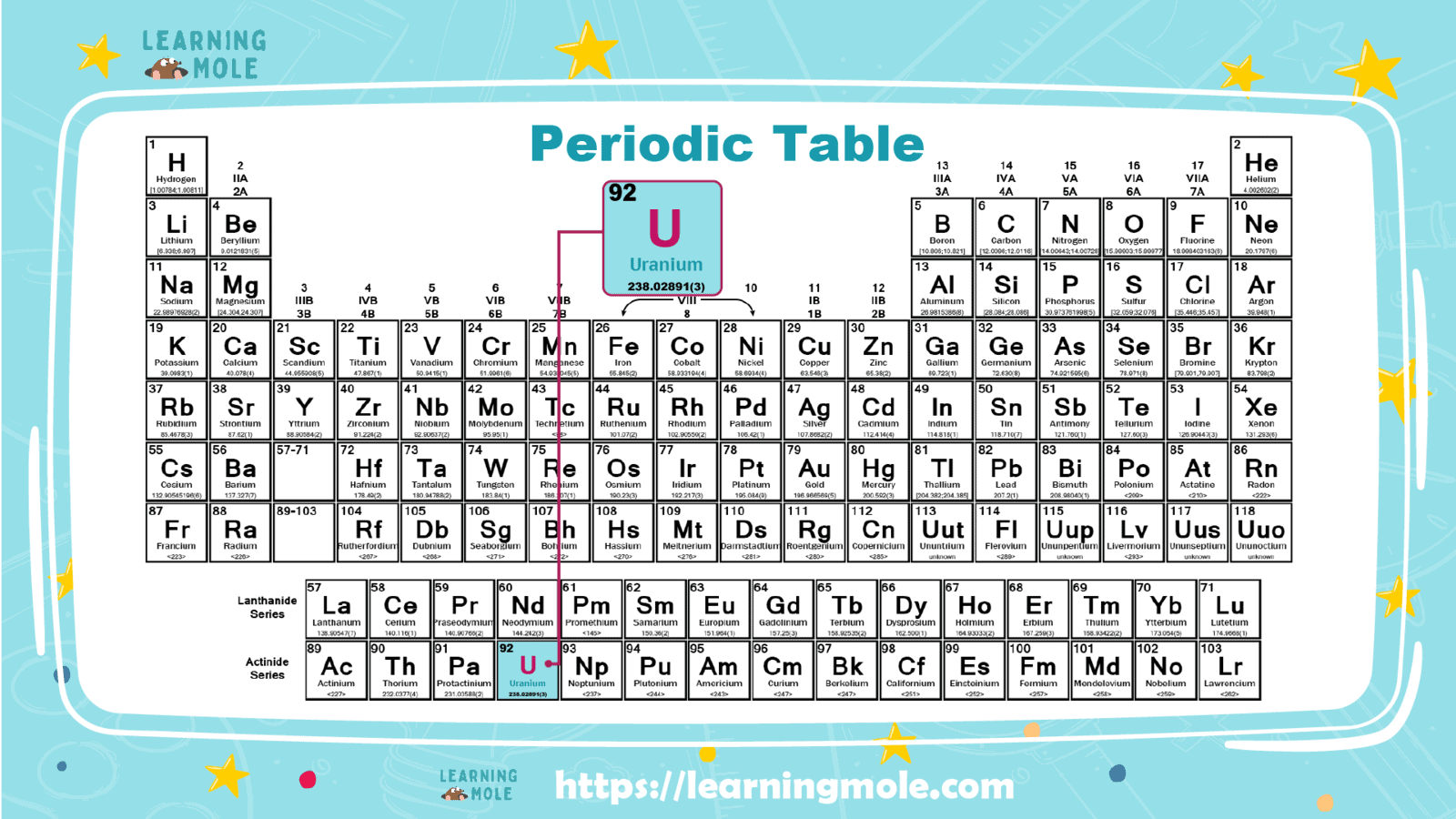
It is an important chemical element, symbolized by the chemical symbol “U”, and its atomic number equals “92”. It is located in the periodic table within the elements of the third group and the seventh period. It is classified into the elements of the lactinide group.
A piece of pure uranium is similar to the element silver or steel, except that its weight is heavier and its density is greater in relation to its size. This uranium atom may contain a large number of protons and neutrons. In addition to that, there are a few of these electrons located in the valence shell.
The uranium element is considered one of the decomposing chemical elements that have high radioactivity, and uranium may occupy the second place in terms of high atomic mass, as it comes directly after the plutonium element, with the need to ensure that it is present in nature before the formation of the earth’s surface, which means that the age of uranium greater than the age of the earth.
The reason behind naming uranium by this name is due to the planet Uranus, which goes back in its name to the Greek god of the sky. In addition to that, uranium was isolated for the first time by heating one of its chlorides with potassium.
A person may be exposed to quantities of uranium by eating a number of foods containing it. In addition to that, he may reach them through breathing; This is due to the fact that it spreads in the air, as these quantities are eliminated through urine or faeces, so these quantities do not pose any danger as long as getting rid of them is relatively easy.

History of Uranium
The history of uranium dates back to the end of the eighteenth century when it was discovered by the scientist Martin Klaproth, and despite its early discovery, its essential properties were not discovered until decades later. Initially, uranium was used primarily as a coloring material, which in turn is used for glazing ceramics and for coloring in early photography, and its radioactive properties known today were not recognized until 1866 AD, and the possibility of using it as an energy source was not evident until the middle of the last twentieth century, as it was used in the aftermath of World War II, by the United States of America, which manufactured Including its nuclear bomb, in which the Japanese Hiroshima massacre was committed, and the matter evolved with it, so that uranium is now used to operate commercial nuclear reactors that produce electricity, and to produce isotopes used for medical, industrial and defence purposes all over the world.
Who discovered Uranium?
Uranium was discovered in 1789 by Martin Kla-Broth, a German chemist, in Pitchblende, in what is now the Czech Republic, when he was analyzing mineral samples in silver mines. The element is named after the planet Uranus, the seventh planet in our solar system.
Regardless of the importance of uranium to chemists, the use of uranium in the nineteenth century was limited to colouring glass and ceramics, as its elements were used to add a greenish-yellow colour to vases and decorative glassware. At the time, it was popularly used to add bright orange to red hues in ceramic glazes used in household pottery and architectural decorations.
In 1896, Henri Becquerel discovered the radioactive properties of uranium. Although he did not realize the importance of his discovery at first, his student Marie Curie realized the results drawn by Henry more accurately and chose the name “radioactivity” for this new phenomenon.
Locations of Uranium
Large amounts of uranium are found in most rocks, in addition to its presence in some types of soil, and there are varying amounts of this element concentrated mainly in water.
Like other elements, uranium may be associated with a large number of different minerals and elements, as it is mainly concentrated in a group of minerals that contain large amounts of uranite, in addition to being present in a number of different minerals such as oranite, atonite, and torbernite.
In addition, uranium may spread in many regions and countries of the world, as it is highly concentrated in the United Arab Emirates in addition to its presence in Saudi Arabia, Algeria and Egypt, and the ores of this element are highly concentrated in both Canada and America.
Uranium isotopes:
Uranium in nature, contains a large number of isotopes, all of which are unstable, starting with the isotope uranium “233” and ending with the isotope “238”, which is considered one of the most stable and radioactive isotopes in addition to its long half-life.
As for the uranium isotope “235”, it may contain a large number of neutrons, the number of which may reach about “143 neutrons”. The counterpart “234”.
The uranium isotope “235” is important in the processes of dating and determining the age of the earth, in addition to being the only isotope that can be obtained in nature. This is because it is considered a fissile isotope that helps the continuation of the nuclear chain reaction.
While the uranium isotope “238” is characterized by the fact that it is capable of fission through the use of a number of fast neutrons, in addition to being a fertile material that is easy to convert into the isotope plutonium “239”, which is vital in many nuclear reactors.
In addition, it is possible to resort to the formation and production of another fissile isotope, which is the uranium isotope “233”, as it is produced mainly from the natural isotope thorium, which is also important in nuclear reactors.

Uranium basic properties
- Uranium is a metallic element with a white colour that often tends to be silvery.
- It is characterized by its heavy weight and great density.
- It appears most often in the solid state.
- Electro-negativity is low compared to the rest of the elements of the group to which it belongs.
- It has a rhombic crystal system.
- It has a magnetic field.
- It can oxidize quickly, especially when exposed to air directly.
- It is characterized by its high radiation.
- Uranium is a toxic element.
- It is one of the most abundant elements present on earth.
- It has its own boiling point.
- This element is of medium atomic mass.

Chemical properties of Uranium
Uranium is an element that occurs naturally in the earth, and like any other element, it possesses its physical and chemical properties, the most important of which are: [2]
- The atomic number: the number of protons in the nucleus, is 92.
- Atomic symbol: On the periodic table of the elements, it is U.
- Atomic weight: which is the average mass of an atom, equal to 238.02891.
- Density: 18.95 grams per cubic centimetre.
- State: At room temperature, it is a solid and has atoms dissolved in water or in air gases.
- Melting point: 1135 degrees Celsius, or 2075 degrees Fahrenheit.
- Boiling point: 4,131 degrees Celsius, equivalent to 7,468 Fahrenheit.
- The number of isotopes: Uranium has 16 isotopes, only 3 of which are found naturally.
- Appearance: Uranium is a heavy, lustrous metal with a silvery-white colour.
- Physically: It is malleable, ductile, and slightly magnetic.
- Chemically: when exposed to air, uranium metal is covered with a layer of oxide, and acids dissolve the metal, but it is not affected by alkalis.
- Impact: Uranium and its compound, uranyl, are highly toxic, both chemically and radiologically.
- Radioactivity: Uranium exhibits three radioactive crystal modifications, alpha, beta, and gamma.
Sources of Uranium
There are several sources of uranium, the most important of which are:
- In minerals, it is found in pitchblende, carnet, cliffite, otonite, uranite, uranovan and torbernite.
- In rocks, uranium is found in phosphate rocks, lignite and monazite sands.
- Uranium can be prepared by reducing uranium halides with alkali or alkaline earth metals or by reducing uranium oxides with calcium, carbon, or aluminium at elevated temperatures.
- The metal can be produced by electrolysis of KUF 5 or UF 4, dissolved in a molten mixture of CaCl 2 and NaCl.
- High-purity uranium can be prepared by the thermal decomposition of uranium halides.
How is uranium extracted?
There is uranium in its raw form extracted from the mine and in a low-grade form, and therefore uranium must be concentrated from these ores to obtain abundant quantities of it. The result is a yellow paste, or the so-called yellow cake, which contains 75% of uranium oxide. After that comes the stage of purifying the yellow dough by a series of chemical transformations and converting it into a fluorinated form, or what is called uranium hexafluoride, so that each uranium atom has one six fluorine atoms, and this is called the conversion process, and thus uranium is a product free of impurities, and ready for enrichment.
What is Uranium enrichment?
In a simple sense, uranium enrichment is a necessary process for creating effective nuclear fuel from uranium extracted from Mother Nature by increasing the proportion of uranium-235, which in turn undergoes fission with thermal neutrons, and the enrichment process is linked to the production process, after purifying the yellow dough and converting it into uranium hexafluoride,
The uranium becomes ready for enrichment through the process of increasing the percentage of uranium-235, from 0.7 percent in natural uranium to about 3 to 5 percent for use in nuclear reactor fuel, and enrichment can be done through several processes, such as the isotope separation process that we explained previously, or Gaseous diffusion or gas centrifuges.[3]
How is Uranium used?
Uranium is used in nuclear power plants to generate heat and heat water to produce steam. Uranium is characterized by its large atoms, which enables it to divide more than other elements.
When non-atomic particles called neutrons to come into contact with uranium atoms, the atoms split apart, releasing thermal energy. This happens all the time in nature, but at a very slow rate. Most nuclear power plants use the isotope Uranium-235, a specific type of uranium, for fuel.
Nuclear reactors are able to significantly speed up this process by slowing down neutrons and making them more likely to collide, which will lead to the fission of uranium atoms. When the uranium atoms split, they release more neutrons, so the process continues, and the divisions increase to ensure a “series of reactions” to divide the atoms. This process is called “nuclear fission.”
Due to the nature of the nuclear fission process, nuclear power plants do not need to burn any type of fuel. Therefore, the plants do not produce any smog or greenhouse gases.
Uranium is used in several fields in addition to its use in power generation.
The uranium-238 isotope, called depleted uranium, is used as a barrier to protect against radiation due to its thickness and density and is often used in the manufacture of aircraft and medical equipment to protect against radiation devices.
Uses of Uranium
Historically, ancient man knew uranium without realizing it when uranium oxide was used as a colouring agent for ceramic coatings, and after its actual and apparent discovery, its most important uses emerged, including:
- It is mainly used in nuclear reactors, in addition to its use in the manufacture of many nuclear weapons and missiles.
- In earth’s geology, scientists believe that uranium and thorium cause most of the earth’s internal heat.
- It can be used in glass colouring operations, With the aim of producing a group of shapes that vary in color from orange to red.
- It is used in photography through its use in coloring and shading.
- Uranium can be used to harden steel.
- It can be used as a fuel for the nuclear energy industry, in addition to being used in the manufacture of some bombs and nuclear weapons.
- It has been used since ancient times in ceramics, by giving it a yellow colour.
- Uranium is used for X-ray targets, which are widely used in the medical field.
- Uranium can be considered a protective shield that protects against all surrounding radioactive materials.
- It is used to estimate the age of igneous rocks in particular. This is due to its half-life and radioactive properties.
- It has been used in some stations due to its excellent ability to generate electricity.
- Uranium-238, with a half-life of 4.51 x 109 years, is used to estimate the age of igneous rocks, which is known as radiometric dating.
- Its nitrate can be used as a photographic powder, and its acetate is used in analytical chemistry.
- It is used in the manufacture and production of many phosphate fertilizers.
- Uranium is used in industries such as guidance devices, gyrocompasses, missile brakes, and others.
- It can be used to desalinate seawater.
- Scientists have suggested that the natural presence of uranium in the soil may be evidence of the presence of radon.
As a result of its heavy weight and high density, it may be used in the manufacture of some aircraft parts, especially the balancing device.
The benefits of uranium
As a natural material, and if it is used peacefully without any other nuclear goals, then uranium is considered one of the most important materials that provide great benefits, especially in the field of energy, and among its most important benefits:[4]
Nuclear energy, produced through uranium, is a carbon-neutral, healthier source of electricity than fossil fuels.
Uranium is a great energy source for reliable primary load power to the electric grid.
It is a carbon-neutral, healthier source of electricity than fossil fuels.
Using less lab-powered materials, such as uranium, means less mining and less impact on the environment.
Uranium is widespread, especially in large industries, and its derived radioactive isotopes are very useful, especially in the medical field.
Uranium, along with some other radioactive materials, is used to know the formation of the earth, its history, and the sources of its underground wealth.
Economically, its production and enrichment are among the industries that develop the economies of the producing countries, especially in Canada, and they also provide tremendous job opportunities.
Uranium damage
Radioactive contamination and heavy metals go hand in hand with the development of uranium. Uranium mining has developed an unfortunate legacy of endangered species, polluting, often permanently, precious public lands and waters, causing catastrophic impacts on human health.
All stages of uranium development, from exploration to mining, milling and production, can pose unique threats to species, ecosystems and human societies, ranging from the destruction of habitat and wildlife disruption to bio-accumulation and irreversible pollution of water and prosperity today threatens to build on the uranium legacy of environmental and social harm.
To this extent, we come to the end of our article, which was entitled What uranium is and its benefits and where is it extracted from, through which we got to know what uranium is, its properties and benefits, the places of its extraction, who discovered it, its history, its sources and its multiple uses, as we learned about its use in the nuclear bomb, and how it is enriched.
Resources
Related Articles
- Copper: Properties, Uses, Reactions and more
- Platinum: All you need to know about the Strong element
- Zinc: Chemical Element Properties, Formation and Uses
- Iron: Properties, Types and Uses


Leave a Reply